Cushing Disease / Cushing Syndrome

Overview
Cushing’s disease is a condition of excessive production of the hormone cortisol due to a benign tumor of the pituitary gland. Common symptoms include high blood pressure, high blood sugar, fatigue, and body fat accumulation to the face, neck, and abdomen.
The wide variety of symptoms and gradual changes in appearance often lead to delayed diagnosis. Although the tumor is benign, symptoms will worsen until treatment is initiated.
Treatment options include surgery, medications, and radiation therapy. Surgery is often curative, is the first line of treatment, and hormone supplementation may be necessary following the procedure. This disease can become life-threatening if left untreated.
What Is Cushing’s Disease?
Cushing’s disease is a condition characterized by an abnormal excess of the stress hormone cortisol due to a benign hormone-producing tumor of the pituitary gland in the brain. This pituitary tumor, also known as a pituitary adenoma, produces excessive adrenocorticotropic hormone (ACTH).
The function of ACTH is to trigger the adrenal glands on the kidneys to produce the stress hormone cortisol. Excessively high ACTH levels leads to an uncontrolled production of cortisol and keeps the body in a constant state of stress. This condition was first described in 1912 by the highly esteemed Dr. Harvey Cushing, who is often considered the founder of modern neurosurgery.
Cortisol production naturally follows a daily cycle or ‘circadian rhythm’. The levels are typically highest in the morning to promote wakefulness and lowest during the night. Cortisol is also produced during situations of acute stress, often known as the “fight or flight” response.
During both cases, cortisol has a wide variety of functions that include regulation of blood pressure, mobilization of energy stores, and suppression of the immune system. However, when this response remains chronically active, disruptive symptoms can occur.
Why should you have your surgery with Dr. Cohen?
Dr. Cohen
- 7,500+ specialized surgeries performed by your chosen surgeon
- More personalized care
- Extensive experience = higher success rate and quicker recovery times
Major Health Centers
- No control over choosing the surgeon caring for you
- One-size-fits-all care
- Less specialization
For more reasons, please click here.
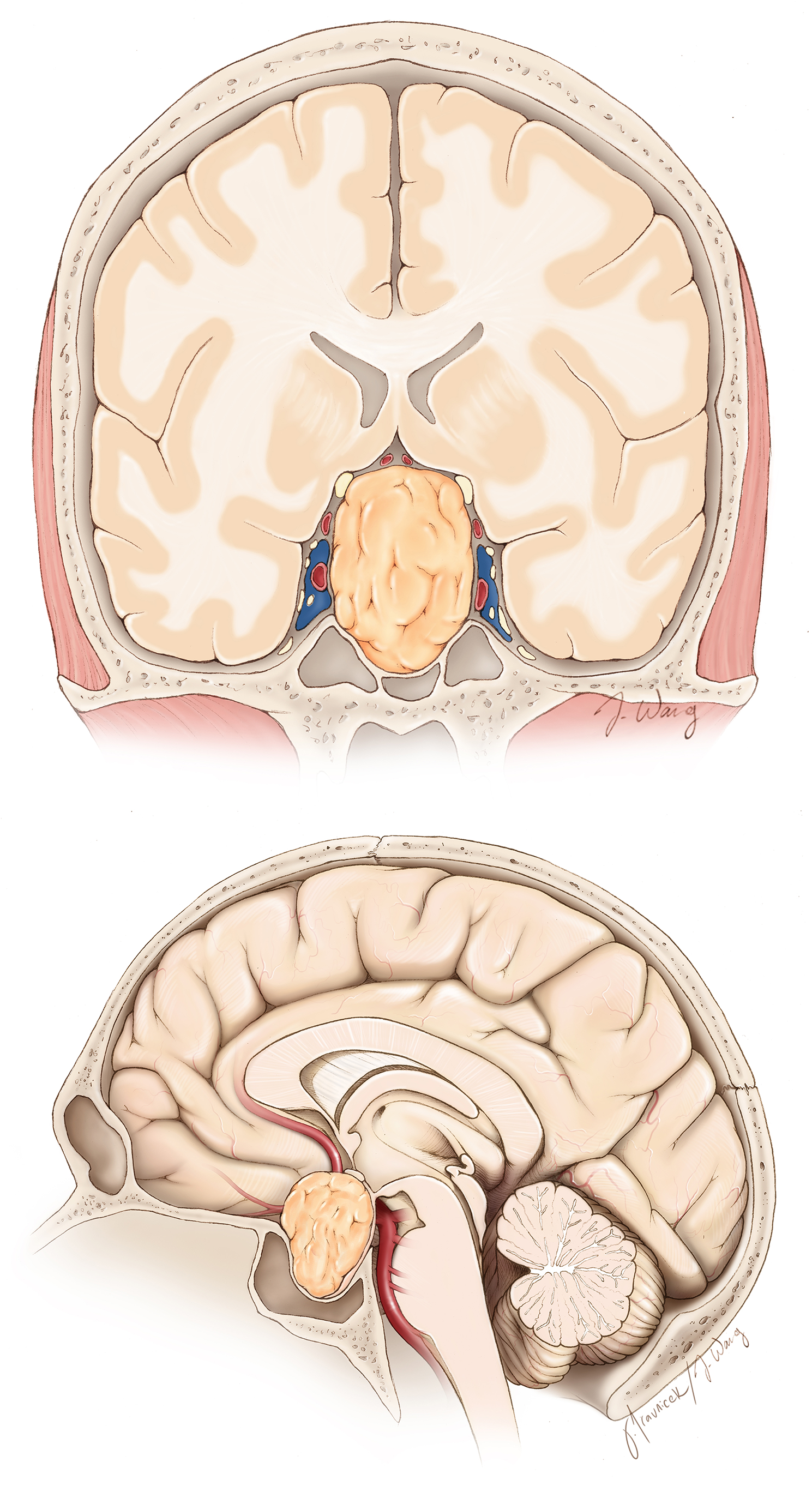
Figure 1. A large pituitary adenoma at the base of the brain is observed compressing the optic nerves and surrounding blood vessels.
What Are the Most Common Symptoms of Cushing's Disease?
Hormones are chemicals that travel in the bloodstream and have effects throughout the body. Therefore, high levels of cortisol in Cushing’s disease often causes a wide variety of symptoms. These symptoms can be subtle and viewed separately, which unfortunately can lead to delayed diagnosis.
General Cushing's disease symptoms can include:
- High blood pressure
- Diabetes mellitus
- Round, fuller appearing face
- Redness of the cheeks
- Fat buildup behind the neck
- Abdominal stretch marks and easy bruising
- Excess hair growth
- Weakness and fatigue
- Muscle loss, particularly of arms and legs
- Abdominal weight gain
- Decreased fertility and sex drive
- Absent menstruation (amenorrhea)
- Mood disorders
- Osteoporosis
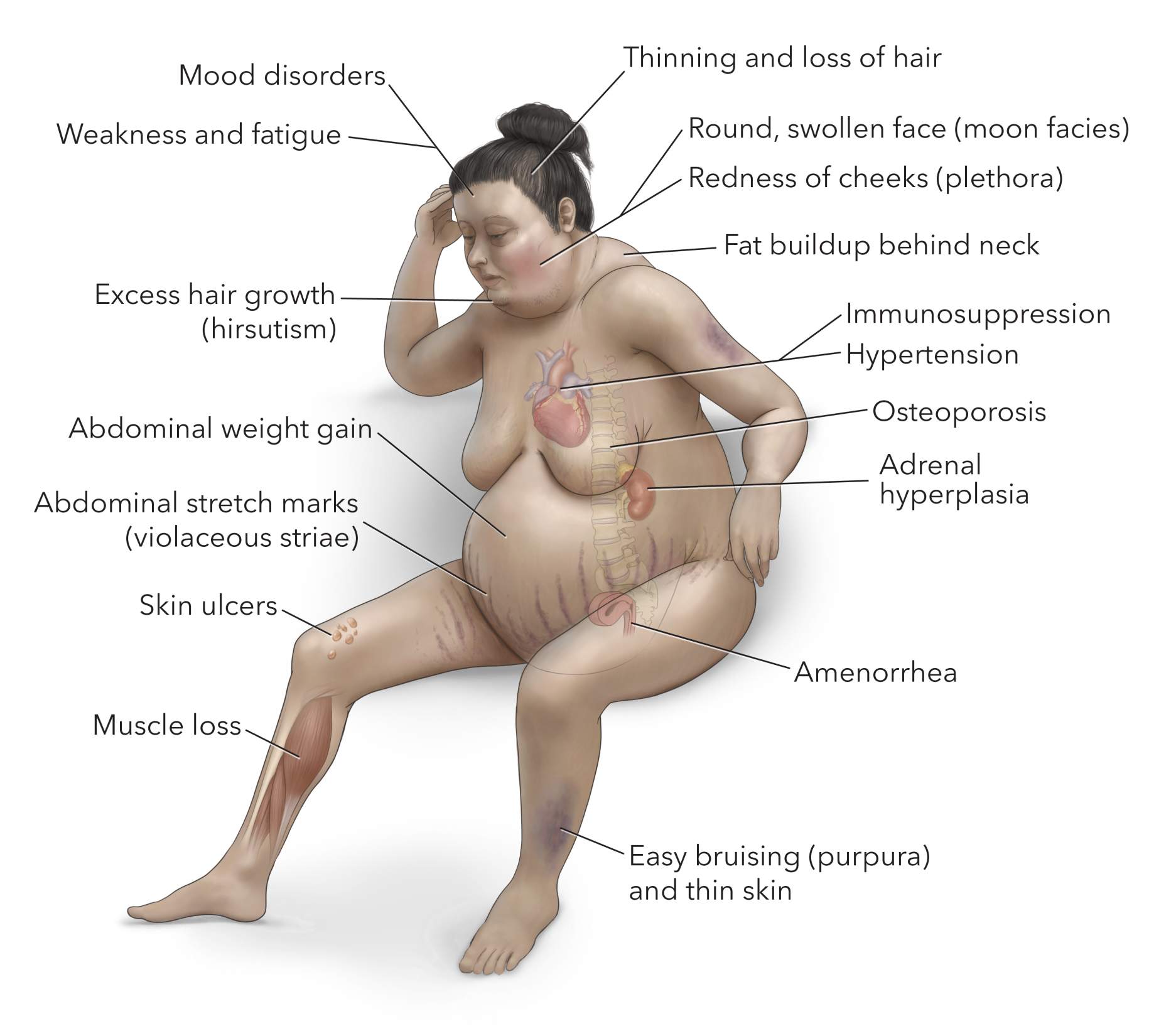
Figure 2. Symptoms of excess cortisol include accumulation of fat behind the neck, abdominal stretch marks, easy bruising, redness of the cheeks, and muscle loss particularly in the arms and legs.
Very large pituitary tumors (macroadenomas) can cause additional symptoms by compressing nearby structures in the head. Due to the proximity of optic nerve fibers, a large pituitary adenoma can cause visual disturbances.
Specifically, a loss of peripheral vision (bitemporal hemianopsia) may be noticed. Vision can become blurry, and colors may appear less vivid.
The pituitary gland is responsible for producing many important hormones in addition to ACTH that affect the release of thyroid, growth, and sex hormones.
If the pituitary adenoma grows large enough to compress the normal pituitary gland, decreased levels of these other critical hormones can occur and lead to symptoms. For example, a reduction in sex hormones could lead to reduced sexual drive, impotence, and infertility.
What’s the Difference Between Cushing's Syndrome and Cushing's Disease?
Cushing’s syndrome (in contrast to Cushing’s disease) is a condition of abnormally high cortisol levels in the body from any source. The most common cause is long-term use of corticosteroid medications such as prednisone, hydrocortisone, and dexamethasone.
This is seen in patients requiring chronic treatment for severe asthma or rheumatoid arthritis. The second most common cause is an ACTH-secreting pituitary adenoma (Cushing’s disease).
Other causes of high cortisol may include tumors of the adrenal glands that secrete cortisol directly, or ACTH producing tumors elsewhere in the body (ectopic Cushing’s syndrome).
Your doctor will conduct tests to rule out these other possibilities during a proper workup for suspected Cushing’s disease. Be prepared to inform your doctor of any current and prior medication use during this evaluation.
What Are the Causes?
Most pituitary tumors occur sporadically without known underlying risk factors or genetic predispositions. Research is being conducted to better understand the development of Cushing’s disease and pituitary adenomas in general.
How Common Is It?
Cushing’s disease is rare with approximately 10-15 cases being diagnosed per million people per year. Most individuals are between the ages of 20-50, with approximately three times as many women than men being affected.
Who Is More Likely To Develop Cushing Syndrome?
Patients of all ages can develop Cushing syndrome, and up to 70% of adults and children develop Cushing syndrome as a result of Cushing’s disease. Some of the risk factors for developing the condition include patients who are:
- Taking cortisol medication for conditions such as asthma or rheumatoid arthritis
- Undergoing long-term therapies with corticosteroids
- Pre-existing condition affecting the adrenal glands
- Pituitary conditions that affect cortisol production
- Pituitary or adrenal gland tumors
How Is It Diagnosed?
The diagnosis is initially based upon a review of the patient’s medical history, physical exam, and laboratory test findings. These will demonstrate the signs and symptoms of elevated cortisol levels and prompt further confirmatory testing such as a 24-hour urinary excretion of cortisol test. Many patients find it helpful to compare current and old photographs as bodily changes are often gradual.
Endocrinologists are primarily responsible for the initial work up and diagnosis of Cushing’s disease/syndrome. Accurate diagnosis can be complex and challenging, especially in mild cases.
Once Cushing’s syndrome is confirmed, the physician will begin further workup to determine the source of the elevated cortisol. The diagnosis of Cushing’s disease can initially be challenging due to the daily fluctuations in cortisol levels and often small tumor size (microadenomas).
The goal of diagnostic testing will be to confirm the presence of an ACTH-producing pituitary adenoma, while ruling out other possible sources of excessive cortisol using the tests described below.
Hormone-Based Testing
The initial goal of laboratory testing is to confirm the presence of elevated cortisol using the following tests.
- 24-hour urinary excretion of cortisol: This test is helpful for confirming a high total level of cortisol from a complete daily cycle. This method is used because cortisol levels can be high or low depending on the time of day. Additionally, this test may be normal in mild cases so multiple tests are sometimes required for diagnosis.
- Late-night salivary cortisol: Cortisol levels are normally lowest at night. In this test, cortisol levels are measured in saliva obtained near midnight. If the levels are elevated, Cushing syndrome is likely. Importantly, if this test is negative, it is very likely that the patient does not have Cushing’s syndrome.
- Low-dose dexamethasone suppression test: This test evaluates the feedback regulation function of the pituitary gland by administering a cortisol-like drug, dexamethasone, which should lower both ACTH and cortisol production.
This is the pituitary gland’s normal mechanism for preventing overproduction of cortisol. If there is an ACTH or cortisol producing tumor present, it will continue to make ACTH and cortisol despite the low dose of dexamethasone.
Therefore, if the test result reveals a high ACTH or cortisol after dexamethasone administration, further workup for the source is necessary.
- High-dose dexamethasone suppression test: This test will likely be utilized if the low-dose dexamethasone suppression test is abnormal. The purpose of this test is to distinguish between a pituitary adenoma or ectopic tumor as the source of elevated cortisol.
A high dose of dexamethasone should be strong enough to suppress a pituitary adenoma from making ACTH, and therefore cortisol.
Levels of ACTH and cortisol are both expected to drop if this is the source. ACTH and cortisol levels will remain high if a tumor of the adrenal glands or other tissue is producing these hormones.
- Pituitary Magnetic Resonance Imaging (MRI): Magnetic resonance imaging is the most detailed method of viewing brain tissues and the pituitary gland. Once obvious causes of high cortisol are eliminated (e.g., medications), an MRI of the head will be obtained to check for a pituitary adenoma.
- Inferior Petrosal Sinus Sampling (IPSS): This is a more invasive procedure (angiogram) where ACTH levels are sampled directly from the veins leaving the pituitary.
These levels are then compared to those of a peripheral vein, which allows the physician to determine if the ACTH source is the pituitary or elsewhere in the body.
This procedure involves a catheter that is placed in the patient’s groin to travel to the veins around the pituitary gland. This procedure is performed as an outpatient and the patient goes home the same day as the procedure.
What Are the Treatment Options?
Cushing’s disease treatment can vary, as patients may need surgery, radiation, medication, or a combination of these therapies, depending on their doctor's diagnosis and recommendation.
Surgery is often the first line of treatment as it is currently the only curative option. For patients who cannot undergo surgery or in cases where surgery is not successful, medication and radiation therapy can potentially manage the condition.
Pituitary Surgery
Endoscopic pituitary surgery is currently the only curative treatment option for ACTH-secreting pituitary adenomas. A minimally invasive surgery can be conducted through the nose using a special instrument with a small camera (endoscope) and the transsphenoidal approach.
During the transsphenoidal approach, surgical instruments are passed through the nose and sinuses to reach the pituitary gland at the base of the skull.
In this video, Dr. Cohen describes the techniques for a sectional focal microadenoma causing Cushing's disease.
For more information about the technical aspects of the surgery and extensive experience of Dr. Cohen, please refer to the chapter on Pituitary Microadenoma in the Neurosurgical Atlas.

Figure 3. Removal of bone through the nasal passages (left) en route to a tumor at the base of the brain (right).
Complication rates are low but can include cerebrospinal fluid leak, excessive urination and thirst (diabetes insipidus), and hormone deficits. These are often temporary conditions. Most hospital stays are no longer than 3 to 4 days.
Some larger tumors usually cannot be removed in the same way. In such instances, the surgeon might opt for a craniotomy. During this procedure, the surgeon exposes the tumor through the upper portion of the skull. This is a more invasive form of surgery, and postoperative stays are longer, usually between 5 and 6 days.
Medications
For patients where surgery is not an option or if surgery has not been successful, the medication pasireotide (Signifor) may be initiated. Pasireotide is a somatostatin hormone analogue that can bind to the ACTH-secreting tumor and inhibit ACTH secretion.
Since this medication can cause blood sugar levels to rise (hyperglycemia), patients with diabetes mellitus should be carefully evaluated and have their current diabetic therapies optimized prior to starting the medication.
Radiation
Radiation therapy is often used after surgery to prevent growth of the residual tumor. If there is evidence of growth of the residual tumor after surgery, radiation treatment might also be considered.
Radiation therapy can come in the form of traditional external-beam radiation or via the more intense and precise stereotactic radiosurgery.
Traditional external-beam radiation therapy directly delivers a single beam of radiation to the tumor. The procedure is usually performed 5 times per week, for several weeks.
The radiation treatments can take several years to bring tumor growth and/or hormone secretion under full control, and the procedure risks damaging normal pituitary function as well as adjacent neural structures.
Stereotactic radiosurgery can deliver higher doses of radiation more accurately by applying precise and multiple beams to target the tumor. It does not require any incision and is done as a single-session outpatient procedure.
As with external-beam radiation therapy, it may take years for the full benefits to manifest, but unlike external-beam radiation therapy, surrounding structures are less prone to damage from the radiation treatment. Treatment is usually avoided for tumors near important nerves.
Radiation therapy can be used rarely as an alternative to surgery for cases in which the tumor is inaccessible via traditional routes.
Possible complications of radiation therapy include headaches, seizures, neurologic problems (depending on the area being treated), and difficulties with thinking or decision-making.
Permanent injury to healthy tissues caused by radiation (radiation necrosis) is another possible but delayed complication of radiotherapy that results in headaches, memory loss, personality changes, and seizures. This complication is rare but can occur months to years after treatment.
Bilateral Adrenalectomy
In severe cases, removal of both adrenal glands located at the top of the kidneys may be required if prior therapy options have failed to cure disease or control symptoms. Without these glands, patients will require lifelong hormone replacement.
What Is the Recovery Outlook?
The recovery outlook for Cushing’s disease depends on the size of the tumor and stage of disease progression. If treatment such as surgery and in the case of small tumors (microadenomas) adequately removes the tumor, patients can expect to return to generally healthy and normal lives.
Pituitary surgery can cure or improve symptoms in 80-85% of patients. Stereotactic radiosurgery is similar in effectiveness with remission rates around 50-60%.
Pituitary adenomas may recur so regular follow up with your physician is important to check for new growths. After therapy, the production of other pituitary hormones may be affected and necessitate hormone supplementation.
Follow-Up Care
Treatment time for Cushing’s disease can vary. During this time, you will need multiple follow-ups with your healthcare provider to report any worsening symptoms.
Aside from symptoms such as weakness and fatigue, recurrence of the tumor can lead to significant physical changes. These can include rapid weight gain, excessive hair growth, acne, and a rounder and redder face. Over time, treatment can potentially lessen these symptoms.
Potential Complications
When left untreated, patients can develop several symptoms that seriously affect their health and quality of life. These conditions can include:
- Deep vein thrombosis (blood clots)
- Compression fractures in the spine
- Diabetes
- High blood pressure
- Kidney stones
- Significant weight gain
Cushing’s syndrome can worsen and be potentially fatal if left untreated.
How Can Cushing's Disease Be Prevented?
Cushing's disease prevention is challenging because it is mainly caused by a benign tumor in the pituitary gland, which is not preventable through lifestyle changes or interventions. Unfortunately, since the underlying cause is not related to modifiable risk factors, there are no specific preventive measures that can be taken to avoid developing Cushing's disease.
However, it is important to remain vigilant and focus on early detection and treatment of the condition to manage symptoms and prevent complications. Regular medical check-ups and being aware of the symptoms can help in the early diagnosis and treatment, which is crucial for a better outcome.
With the right care and attention, it is possible to manage the condition effectively.
What Can Patients Expect in Terms of Quality of Life After Diagnosis/Living With Cushing's Disease?
After diagnosis and treatment of Cushing's disease, patients can experience a significant improvement in quality of life, particularly if the disease is identified and treated early.
Successful treatment can alleviate many of the physical symptoms (e.g., weight gain, muscle weakness, and skin changes) and psychological symptoms (e.g., depression, anxiety, and cognitive difficulties) associated with the disease.
However, the extent of improvement can vary depending on factors such as the duration and severity of the disease prior to treatment, the type of treatment received, and the presence of any complications or comorbidities. It's important for patients to have regular follow-up care to manage any ongoing or emerging health issues and to monitor for potential recurrence of the disease.
What Is the Cushing’s Disease Life Expectancy For Patients?
Living with Cushing’s disease can be a challenging experience that involves a range of factors, including the cause of the condition, the severity of symptoms, and the effectiveness of treatment.
However, with appropriate management and treatment, many individuals can lead fulfilling lives with a near-normal life expectancy. Treatment typically aims to normalize cortisol levels and may involve tapering off or stopping corticosteroid medications, surgical removal of tumors, medical therapy, or radiation and chemotherapy if surgery is not possible.
Resources
Glossary
Adrenocorticotropic Hormone (ACTH)—hormone produced by the pituitary, which causes the adrenal glands to produce cortisol
Adenoma—benign tumor of tissue containing glands
Benign tumor—tumor that remains in its primary location without spreading
Cortisol—hormone secreted by the adrenal glands that activates the stress response and alertness
Craniotomy—procedure that removes a piece of bone from the skull to expose the brain; the bone is reattached to its original location immediately after the rest of the surgery
Diabetes insipidus—salt and water processing abnormality that leads to excessive urination and thirst caused by problems with antidiuretic hormone
Pituitary gland—pea-sized gland at the base of the brain which regulates many important hormones
Radiation necrosis—injury of healthy cells caused by radiation
Contributors: Bradley Estes BS, Gina Watanabe BS
References
- Chung Thong Lim, Márta Korbonits Korbonits, Update on The Clinicopathology of Pituitary Adenomas, Endocrine Practice, Volume 24, Issue 5, 2018, Pages 473-488, ISSN 1530-891X, https://doi.org/10.4158/EP-2018-0034.